Clinical Research Center
Overview
The Clinical Research Center of Nagasaki Medical Center started in April, 1978 as the Clinical Research Department. On October 1, 2002, with new organization and facilities, it was re-named the Clinical Research Center, with the goal of enlarging the hospital’s capacity as a National Hospital for liver patients.
Learning from patients for the benefit of patients.
- Pursue genuinely valuable research.
- Provide the highest level of specialized care for liver patients.
- Improve the quality of treatment and raise up clinicians for liver patients.
- Disseminate information and research results on liver disease to the world.
- Provide medical care, research, and international support from an international perspective.
Within the field of liver disease, this center puts particular strength into the following areas:
- Viral hepatitis / liver cancer
- Incurable liver diseases (such as autoimmune hepatic syndrome)
- Improving the quality of treatment for liver patients
- A facility with the highest level of specialized care for liver patients.
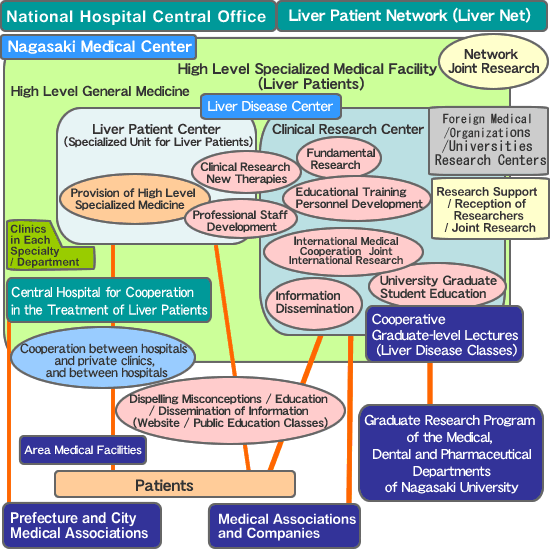
As one of the areas of medicine that should be pursued by this nation, this hospital strives to be on the forefront of diagnosis and treatment technology for liver patients, working to improve capability in treatment, clinical research, dissemination of information, and education, establishing a position as a high level specialized medical facility.As part of our position of strengthening research, we established the Clinical Research Center, with fifteen laboratories in five sections.Each research section has five full-time research section chiefs and laboratory heads (five full-time). Researchers are deployed to the various laboratories, and while pursuing their independent research they maintain communication with the other sections. - Liver Patient Center (a dedicated unit to provide liver patients with the highest level of specialized medicine)
As a place for applying the highest level of treatment to liver patients, the Center incorporates 90 beds on the 8th floor of the inpatient building, serving liver patients as well as digestive tract patients. This unit has a dedicated laparoscopy room and an ultrasonic testing and treatment room. Specialist doctors and nurses are involved in patient care. In addition to the Digestive Tract Group we also have such facilities as Digestive Tract Surgery, Radiology Unit, Emergency Center, Radiology Department, Research Testing Unit, Pharmacology Department, and Nutrition Department, with close cooperation among the different sections. Because of this, patients can be rest assured of receiving full treatment on all levels. A Liver Disease Class is held once a month, enabling the inpatients to gain knowledge and understanding of their problems. - Strategic Medicine Liver Disease Network
As a member of the National Hospital Liver Disease Network (Liver Net), we have amassed a great deal of evidence to promote EBM that should be shared around the world, and through this network we are promoting collaborative research. Liver Net is made up of 13 liver specialty facilities and 14 cooperating facilities. In addition to viral hepatitis/liver cancer and incurable liver diseases, which are of particular concern for the Japanese people, we are pursuing research to improve the level and quality of medical care. - Clinical research
With research funding supplied by the Academic Advancement Committee of the Culture Ministry and the Liver Disease Research Group of the Welfare and Labor Ministry (the Liver Disease Research Group supported by the Welfare and Labor Ministry Scientific Research Fund), we have organized and, along with researchers across the nation, are conducting research to support the liver disease program encouraged by the national government. - Implementing New Therapies
Aiming for smooth operation of the research with which we have been entrusted, we are designing new therapies around the research on the various liver diseases and the care of patients with those diseases, coordinated by the Therapy Management Office.We are aiming to enlarge CRC operations and improve our standing (with certified personnel), since CRC should be the model for a therapy management office and therapy operations. Maintaining close association with our locality, and wanting to keep the door wide open for citizens to receive new therapies, by maintaining close communication with those undergoing the therapies, we strive to perform new therapies with broad understanding by the general public. We also conduct training and education in CRC.We are very proud of the fact that even within the National Hospital organization, we have established a track record of the very high rate of 90% of our treatments being implemented. - A Graduate Program of Nagasaki University (Cooperative Lectures)
Since 2004 we have been holding cooperative lectures with the joint graduate research program of the medical, dental and pharmaceutical departments of Nagasaki University. Nine teachers (six professors and three assistant professors) teach on liver diseases to graduate students from the Response to New Diseases program. It is the first time in this nation for a hospital to be designated in this way for a university graduate studies program. - The Central Hospital for Cooperation in the Treatment of Liver Patients
In August, 2007, we led the nation by receiving this designation from Nagasaki Prefecture.We disseminate information as the central hospital for Nagasaki Prefecture for treatment of liver patients, focusing particularly on treatment, support, and counseling of viral hepatitis patients. - International Cooperation in Medicine
We are very active in hosting trainees and researchers from other nations. In 2004 they came from Drum Tower Hospital of Nanjing University Medical Department in China, in 2006 they came from Bei Hua University, also in China, and in 2007 they came from the Scientific Research Institute of Cardiology and Internal Diseases of Kazakhstan (abbreviated as the Kazak Cardiology Center). Through such things as a 2-day JICA conference on viral hepatitis, our participation in international training programs is constant. Also, as a part of Nagasaki Prefecture’s Welcome Internationals Project, we have welcomed one or two Chinese trainees at a time, giving them guidance primarily through the Clinical Research Center. In 2005 we opened the Overseas Travel Outpatient Clinic, providing consultation and vaccinations for those planning overseas travel, something so unique that this year the Japan Economic Newspaper did an article on it.Previously, things involving international cooperation in medicine have been handled by the International Medical Cooperation Office, but from the need for the whole hospital to be involved, in such things as nurses’ training and involvement, in 2005 we established the International Medical Education Research Center, which works in cooperation with the International Medical Cooperation Office.
History
| 2007 August | Hospital was designated as the central hospital for treatment of liver patients. |
|---|---|
| 2007 June | Reorganization of Liver Net and reestablishment of cooperative research. |
| 2006 April | Synthesis of the JICA Kenya KEMRI project |
| 2005 June | Started the Cirrhosis Research Group (Second Yatsuhashi Group) under the Welfare, Labor, and Science Ministry |
| 2005 March | Completion of the L-net electronic registration system (Continued from 2000) |
| 2004 June | Began the Acute Hepatitis Research Group on Liver Net and received the Viral Hepatitis Foundation Encouragement Award (Organization Division) |
| 2004 April | Began the “Liver Disease Classroom” in the Liver Patient Center |
| 2004 April | Designated a Graduate Program of Nagasaki University (Liver Disease Lectures) General Medical, Dental and Pharmaceutical Research Department Liver Disease Lectures of the Response to New Diseases Program |
| 2003 April | Started the PBC Treatment Method Research Group (Ishibashi Group) under the Welfare, Labor, and Science Ministry |
| 2002 October | Began operation of the Clinical Research Center |
| 2002 April | Started the Hepatitis C Research Group (Yatsuhashi Group) under the Welfare,Labor, and Science Ministry |
| 2001 September | Opened the Liver Patient Center (90 beds) |
| 1999 March | Designated an Associate National Center by the Liver Disease Hospital Association |
| 1991 April | Practical operation of an electronic medical records system |
| 1987 April | Started development of an electronic medical records system |
| 1981 April | Designated a WHO Viral Hepatitis Center |
| 1978 April | Established the Clinical Research Department |
| 1976 December | Opened a Liver Patient Unit (50 beds) |
| 1975 April | Designated a Facility for Liver Patients (Under the Facilities Plan of the Welfare Ministry) |
Research Structure
This hospital, as a part of the medical policies of our nation, focusing on cutting-edge therapies and diagnosis for liver patients and incurable diseases, aiming to improve treatment techniques and capability, has established a place as a top level specialized medical facility in treatment, clinical practice, research, dissemination of information, and education.
The Research Department is made up of the Treatment Research Section, the Incurable Disease Research Section, the Disease Cause Analysis Research Section, and the Functional Form Research Section, for a total of 13 laboratories. Each research section has four full-time staff members, including a Section Chief and laboratory chiefs and researchers. These maintain communication and cooperation with each other while pursuing independent research.
| Research Department | Laboratory |
|---|---|
| 1.Treatment Research Section | Hepatitis Treatment Research Lab |
| Liver Cancer Treatment Research Lab | |
| Liver Failure Treatment Research Lab | |
| New Therapies Management Office | |
| International Medical Cooperation Office | |
| 2.Incurable Disease Research Section | Disease Status Control Research Lab |
| Genetics Research Lab | |
| Information Research Lab | |
| 3.Disease Cause Analysis Research Section | Virus Immunity Research Lab |
| Autoimmune Research Lab | |
| 4.Functional Form Research Section | Imaging Research Lab |
| Surgical Treatment Research Lab | |
| Pathology Research Lab |
Areas of Individual Research
-
Viral Hepatitis
- (1)Acute Hepatitis (New and old infection types, epidemiology)
- (2)Chronic Hepatitis and Cirrhosis (Preventing liver cancer onset)
- (3)Liver Cancer (Treatment, cancer onset mechanisms)
-
Incurable Liver Diseases
- (1)Autoimmune Hepatic Syndrome (Cirrhosis caused by bile from radiation)
- (2)Autoimmune Hepatitis
-
Improving the Quality of Medical Care
- (1)Evaluating and Qualifying Clinical Care
- (2)Clinical Care Guidance